Latest News

A new study found that ZetrOZ Systems’ sustained acoustic medicine (sam) and other low-intensity continuous ultrasound technology, alleviated the symptoms of chronic lower back pain and reduced the need for pain medication.

ZetrOZ Systems, the creators of sustained acoustic medicine and the manufacturers of the revolutionary sam® wearable ultrasound unit, concluded the first half of 2024 with presentations at three major medical conferences. At these events, the company shared the latest research on the effectiveness of its technology in enhancing platelet-rich plasma treatments for soft tissue healing.

ZetrOZ Systems, the creator of sustained acoustic medicine, will be participating in the College Athletic Trainers’ Society (CATS) 2024 Symposium to showcase its sam® wearable ultrasound unit. This pioneering device is designed for injury healing. The conference will take place on May 15-18 at The Orleans Hotel in Las Vegas, and ZetrOZ Systems will be at Booth 29. CATS was established to promote quality care in intercollegiate athletics.
The US Food and Drug Administration (FDA) has granted clearance to ZetrOZ Systems’ sustained acoustic medicine device, the sam 2.0 wearable ultrasound unit, broadening its use for various medical conditions.

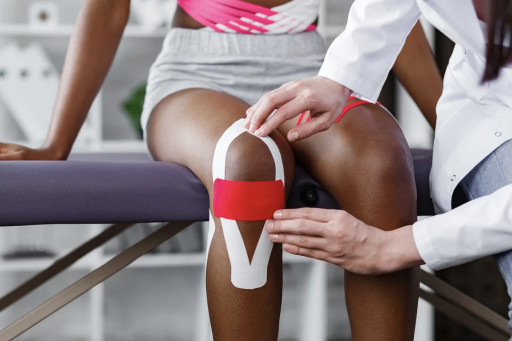
US-based ZetrOZ Systems has received expanded indication approval by the US Food and Drug Administration (FDA) for its sam 2.0 long-duration wearable ultrasound unit to treat soft tissue injuries and knee osteoarthritis.

If you've ever experienced a running injury (or had a baby), you're probably familiar with ultrasound as a diagnostic imaging tool. For years, ultrasound has also been used as a therapeutic tool, and recent advances in technology have made it possible to miniaturize the technology for home use, at much lower intensity and for longer duration than it is used in a clinical setting. The research is not extensive, and it's not cheap. But some runners have found it to be the thing that works where nothing else does.

We have designed sustained acoustic medicine (sam®), a wearable ultrasound device that reduces the need for surgery and pain medication, and can be used by the patient during daily activities. The sam® family of products are the only wearable long-duration mobile ultrasound therapy devices approved by the FDA to provide therapy for up to four hours daily.

