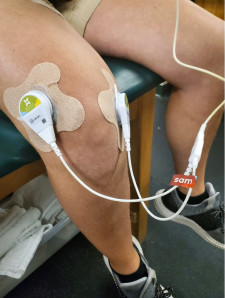
Wearable SAM ultrasound treatment is shown to be a cost-effective solution for the treatment of knee arthritis pain, compared to physical therapy and hyaluronic acid injections.
TRUMBULL, Conn., November 17, 2020 (Newswire.com) - ZetrOZ Systems, developers of Sustained Acoustic Medicine (SAM), a wearable, home-use, FDA-cleared bio regenerative medical device, announces that its product has been recognized as a cost-effective treatment for patients suffering from knee pain relating to arthritis in a recent research study published in the Journal of Orthopaedic Surgery and Research. Over 60 million Americans suffer from arthritis-related pain and disability. For those seeking non-surgical and non-invasive treatment methods for reducing pain while improving function and quality of life, SAM is a promising alternative demonstrating improved clinical outcomes for patients.
Dr. George Lewis, CEO of ZetrOZ Systems, said, "We're glad to be able to offer a treatment solution that offers increased effectiveness in both efficacy and cost. ZetrOZ is committed to providing conservative treatment options to patients looking to achieve pain relief and functional gains without the need for surgery. The use of SAM has been adopted by many government healthcare networks such as the Veterans Administration and Department of Defense, and we look to expand access to all Americans in the private sector."
A 2020 study published in the Journal of Orthopaedic Surgery and Research concluded that sustained acoustic medicine is a safe, effective, non-surgical and non-opioid treatment option for patients with arthritis pain. The study concluded that, "Based on accepted thresholds in determining the cost-effectiveness of therapies in treating knee osteoarthritis (OA), SAM, wearable home-use continuous long-duration ultrasound, appears to be a cost-effective therapy and should be considered when treating patients with knee OA."
Research and funding for the use of Sustained Acoustic Medicine for the treatment of knee arthritis pain are funded and supported by the National Institutes of Health.
About ZetrOZ Systems
ZetrOZ Systems is an FDA cGMP and ISO 13585 medical technology company headquartered in the southern coastal region of Connecticut. The organization also has manufacturing facilities across the United States. ZetrOZ Systems produced UltrOZ®, sam®Sport and sam®Pro 2.0 to provide safe and effective treatment options for prevalent conditions such as arthritis. Learn more at zetroz.com and samrecover.com.
Media Contact: melissapr@newswiremail.com
Source: ZetrOZ Systems
Share:
